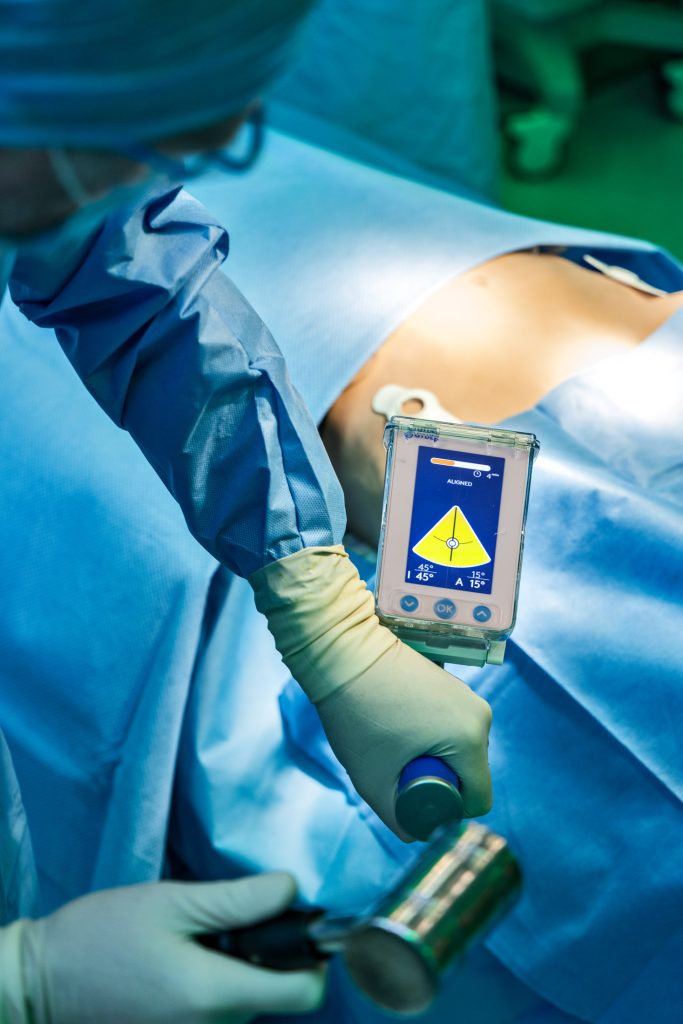
The GYDER® Hip Navigation System is a pin-less, computer assisted surgical guidance system that provides real time guidance during Total Hip Arthroplasty using the Anterior Approach.
MULGRAVE, Australia & Palo Alto, USA, Jan 3, 2024 (GLOBE NEWSWIRE): GYDER® Surgical, a MedTech company specializing in intuitive, non-invasive orthopaedic navigation solutions, today announced the successful completion of India’s first total hip replacement surgeries using the GYDER® Hip Navigation System. The cases were performed in Nov 2023, by Dr. Supreet Bajwa, a fellowship trained orthopedic surgeon and a leader in the Anterior Approach for Total Hip Arthroplasty.
Surgeries using the system were performed at two leading hospitals, Narayan Hrudalaya and the Sanjay Gandhi Orthopaedic and Trauma Hospital, in Bengaluru, India. The life changing surgeries showcased the transformative capabilities of the pin-less navigation system. Unlike traditional methods, this cutting-edge patented technology provides surgeons with real-time guidance to correctly position and implant the acetabular cup without the need for invasive metallic pins. Developed by GYDER® Surgical, the innovation does not rely on pre-operative or intra-operative imaging such as CT ( Computer Tomography ) or X-ray technology for anatomical landmark registration. One minute calibration and quick registration makes the GYDER® Hip Navigation System a fast, easy-to-use, seamlessly integrated computer assisted navigation solution. Improving cup placement accuracy is clinically important and has significant potential to reduce post-surgery complications, thereby improving patient outcomes and lowering health system cost burden.
Dr. Supreet Bajwa expressed excitement about the potential impact of the GYDER® Hip Navigation System, “In my practice, the GYDER® Hip System represents a significant leap forward in navigated hip surgery and stands as a beacon of precision and positive outcomes. The elimination of invasive pins, and minimizing the need for additional intraoperative imaging, makes this system remarkably efficient, allowing for a quicker and more surgeon and patient friendly surgical experience. Coupled with the Anterior Approach, we embark on a surgical journey where meticulous accuracy, simple workflow, and transformative experience converge, shaping the future of orthopedic excellence in India”.
“The first successful use of the GYDER® Hip System in India represents an important milestone in bringing this solution to a large emerging market that needs accessible technologies to address the growing patient demand for orthopedic surgeries. We believe that the best solutions minimize complexity and do not require surgeons to significantly change their practice. And the GYDER® Hip System does exactly that.” Sujit Dike, CEO, GYDER® Surgical.
GYDER® Surgical received Australia’s TGA regulatory approval in 2023 and has recently been granted human research ethics committee approval to undertake a Post Market Study in Adelaide, Australia. The study is scheduled to commence in January 2024 and is expected to further validate the efficacy of the pin-less navigation system in real world practice. GYDER® Surgical plans to submit a 510(K) application to the US FDA in 2024. GYDER® Surgical was also recently awarded a Grant Award – GA340141 through the Entrepreneurs’ Programme – Accelerating Commercialisation under the Australia Department of Industry, Science and Resources to commercialize its product in Australia.
About Gyder Surgical
Gyder Surgical is a MedTech company specializing in intuitive, non-invasive orthopaedic navigation products. We believe that every patient should have access to accurate and precise surgery, no matter where they are. We are building products that work in a wide variety of care settings – Ambulatory / Outpatient Surgery Centers or large ORs in hospitals. The GYDER® Hip Navigation System, our first product, is designed to assist surgeons to accurately place the acetabular cup during hip surgery
For media inquiries or further information, please contact: pr@gydersurgical.com
Website: https://www.gydersurgical.com
LinkedIn: https://www.linkedin.com/company/gyder-surgical/